Hypoxic respiratory failure in an air ambulance patient

Drs Jon Warwick and Neil Crooks discuss hypoxic respiratory failure and the effects of cabin pressurisation on patients with respiratory conditions, considering whether or not patients with high oxygen concentrations may benefit from a lower cabin altitude during transfer
Introduction
How do patients with hypoxic respiratory failure behave when exposed to the reduced ambient pressure of the aircraft cabin? For reasons of sensible aircraft engineering, the cabin of a typical jet aircraft at cruising altitude is pressurised to approximately 80 per cent of its sea level value; the partial pressure of oxygen in the cabin will decline similarly. The cabin pressure may decrease from a mean sea level value of 101.3 KPa towards 80 KPa (a 20-per-cent reduction) at an effective cabin altitude of some 5,000 feet. For routine patients needing supplemental oxygen when in the hospital ward, a modest increase in their inspired oxygen fraction (FIO2) when in the aircraft cabin is all that is required to offset this reduced ambient pressure. This can be delivered by simple means (facemask or nasal cannula) to restore adequate oxygenation, and the effects monitored via pulse oximetry (SpO2). If not breathing by themselves, patients requiring artificial ventilation or support may need a higher FIO2 delivered by the ventilator.
Patients already receiving maximum FIO2 that we may meet in intensive care provide us with a more significant challenge; these patients are the main focus of this article. Clearly, patients should be in the best possible condition prior to transfer and a high FIO2 might indicate room for improvement, for example the treatment of infection. But if delay is not possible, and the reason for transfer is to access a higher level of care, then clearly one must consider the effects on oxygenation of cabin pressure during flight. If required, any cabin pressure limitation may incur penalties of reduced flight level, aircraft endurance and range, and increased total transfer time.

Clinical observations
The response of patients in severe respiratory failure during transfer is not always predictable. We have observed patients being treated for profound hypoxia, receiving 100 per cent inspired oxygen at sea level, whose arterial oxygenation was changed very little in the aircraft cabin with standard pressurisation. For example, a patient whose indication for transfer was to an advanced respiratory centre to receive extra-corporeal membrane oxygenation (ECMO) (Fig. 1) had SpO2 values that remained unchanged at normal cabin altitude. But we have also transferred similar critically ill patients who have experienced a significant fall in SpO2 despite comparable flight conditions. A descent of the cabin was required to increase ambient pressure, and hence increase inspired oxygen partial pressure (PIO2) whilst maintaining the same maximal FIO2. This article will explain why this is so.

Normal physiology
The oxygenation of blood and the removal of carbon dioxide at the alveolar capillary interface is an extremely efficient process. Total surface area for gas exchange in the adult depends on body size; the true value is a matter of debate, but there may be some 500 million alveoli with a total surface area of 150m2. The diffusion distance across the blood/gas interface is just 1 m, and in some places even thinner. Transit time for an erythrocyte to traverse the alveolar capillary (i.e. from the venous to the arterial side of the vasculature) is 1 sec, and the haemoglobin (Hb) it contains will become fully saturated with oxygen in approximately the first quarter of its journey (¼ sec) along the capillary. Blood flow in the lung (Q), and ventilation (V), must be closely matched (V/Q 0.8) for optimum gas exchange to occur, and there are various mechanisms within the lung that help to achieve this.
Arterial oxygen partial pressure (PaO2) is related to both content (CaO2) and haemoglobin saturation (SaO2, or SpO2 if measured by pulse oximetry) by the oxyhaemoglobin dissociation curve (Fig. 2). Normal PaO2 is approximately 13.3KPa, and assuming a Hb concentration of 150 g/l, and normal oxygen carrying capacity of 1.34 ml/g, this will produce a CaO2 of some 200 ml/l (or 20 ml/dl). Normal cardiac output is 5 litres/min, giving a total oxygen delivery (from left ventricle to aorta) of 1,000 ml/min.


Abnormal physiology
The leading cause of hypoxic respiratory failure in patients receiving intensive care is ventilation/perfusion (V/Q) mismatch. This is true for a wide range of conditions, for example pulmonary oedema, pneumonia, sepsis, aspiration, blast injury – indeed, almost every cause of acute respiratory distress syndrome (ARDS). Following an injury or insult from whatever cause, the lung responds by becoming wet, inflamed, poorly-aerated, and stiff. These patients have areas of the lung that are inadequately ventilated in comparison to their perfusion (V/Q<<1). This low V/Q area will result in arterial blood with a depressed oxygen content. Blood from these lung areas will mix with blood from areas with normal and higher V/Q ratios, and hence normal or elevated PO2 values. What is sometimes not appreciated is that even if blood from low V/Q regions (with low PO2) mixes with blood from high V/Q regions (with high PO2), the resulting mixed arterial blood will always have a depressed PO2 and SaO2. This is because it is the oxygen contents from these different areas that become averaged in the pulmonary vein, and not an average of PO2. High V/Q regions cannot increase their oxygen content despite their elevated PO2. Oxygen content is primarily due to the oxygen bound to Hb; this is practically maximal even at a ‘normal’ PO2.
Shunt refers to that fraction of the pulmonary blood flow which effectively bypasses the gas-exchange surface
Shunt refers to that fraction of the pulmonary blood flow which effectively bypasses the gas-exchange surface. Pure shunt (V/Q=0) is clearly not correctable by an increase in FIO2, since both ventilation and perfusion are completely separated – no amount of increase in alveolar partial pressure of oxygen (PAO2) can compensate for a situation where there is no ventilation to a perfused alveolus. True shunt is usually small. In sick patients with ARDS-type respiratory failure, the depression of arterial oxygenation is principally due to V/Q mismatch rather than shunt, but since it is not easy to quantify degrees of V/Q mismatch, it is conventional to describe it in terms of the degree of true shunt which would produce the same observed gas exchange abnormality, hence the term ‘shunt-like effect’.


It is important to understand the relationship between FIO2 and PaO2 (Fig. 3). In health, with effectively zero shunt-like effect, there is an almost direct linear relationship between the two. As FIO2 increases, then PaO2 will also increase. But as shunt fraction increases in patients with V/Q inequality, then a progressively higher inspired oxygen percentage is required to achieve a given PaO2. It can be seen from Fig. 3 that as shunt fraction exceeds 25 to 30 per cent, then large changes in FIO2 can be made with only minimal improvement in PaO2. To put it another way, arterial oxygenation (measured by content, partial pressure or haemoglobin saturation) becomes progressively independent of the inspired oxygen percentage above shunt fractions of 25 to 30 per cent. In effect, the patient becomes increasingly ‘oxygen resistant’. And it follows that the more oxygen resistant a patient becomes, then the less their oxygenation will be affected by changes in ambient pressure, i.e. aircraft cabin altitude. This is the explanation why we sometimes meet patients on intensive care, receiving high inspired oxygen concentrations, whose oxygenation remains much the same despite the reduction in ambient pressure of the aircraft cabin.
Do all patients behave like this?
In contrast, we also meet patients who behave differently. Despite being ventilated with 100 per cent oxygen, some patients are exposed to an unacceptable decline in oxygenation as a result of this reduced ambient pressure (and hence reduced PAO2). This might be because their particular V/Q mismatch and degree of shunt-like-effect is at a level where changes in inspired oxygen partial pressure do indeed make a significant difference; however, there may be a different explanation. V/Q mismatch, although the most common reason for hypoxic respiratory failure, is not the only reason. Some patients may have failure of oxygenation due to an abnormal alveolar diffusion barrier. Patients with interstitial lung disease, such as idiopathic pulmonary fibrosis, have abnormal diffusion and can be extremely sensitive to reductions in PAO2.
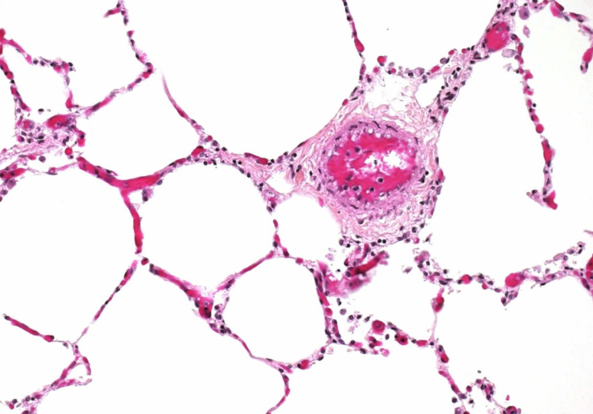
The problem of impaired oxygen diffusion
Pulmonary fibrosis is characterised by an abnormal thickening of the alveolar wall (Fig. 4), and the lungs become stiff and increasingly scarred. The equilibration between alveolar gas and blood that normally takes ¼ second, and is accomplished within the first 25 per cent of blood transit around the alveolus, now takes much longer. As the disease progresses, this equilibration process may require a greater distance (and time) along the alveolar capillary to be complete. Ultimately, a state will be reached whereby equilibration is still incomplete by the time the erythrocyte reaches the end of the alveolar capillary. These patients now become dependent on supplemental oxygen.
The role of the delivered oxygen is to raise PAO2, and so raise the partial pressure gradient to increase the speed of diffusion (Fig. 5). Any reduction in PAO2, for example due to the reduced cabin pressure in flight, must be counteracted with an increased FIO2. If FIO2 cannot be increased further, then SpO2 must fall. If the resulting SpO2 and reduced oxygen delivery is considered to be unacceptable, then the only treatment option remaining is to lower the aircraft cabin (i.e. raise cabin pressure). Some patients can indeed be very sensitive to the effects of cabin pressure changes.

How do I manage my patient?
Patients receiving significantly less than 100 per cent O2 at sea level (for example, <60 per cent) are generally straightforward to manage since inspired concentration can simply be increased in flight. CPAP or NIV for spontaneously breathing patients might provide an additional strategy, otherwise intubation and ventilation for transfer may be indicated. It is the patients on higher concentrations who are the challenge, because there is less room for manoeuvre.
We can gain a clinical impression whilst in the intensive care unit about how critically oxygen-dependent a patient may be: simulate the effects of an aircraft cabin by applying a 15-20 per cent reduction in FIO2 and observe (with repeat arterial blood gases) how the patient behaves. This will help to plan your strategy. Patients who profoundly desaturate are not going to cope well ‘at altitude’, and you may need to plan for a cabin pressure limitation if transfer is to go ahead.
In everyday clinical practice, it can be difficult to predict how the patient will cope in flight and you may need to just go and find out. A ‘stepped climb’ rather than a continuous climb to cruise altitude will help provide extra time for patient assessment as cabin pressure reduces (and altitude rises). Request the flight crew maintain a slow rate of cabin climb (rate of change of cabin pressure is selected low) whilst you continually monitor the patient. The pre-flight briefing between flight crew and medical crew must address any contingencies that might be needed as a consequence of any cabin pressure limitation that may need to be implemented, such as an extra technical stop, or change to flight planned route.
