Prone position in intensive care transport – possible or no-go?
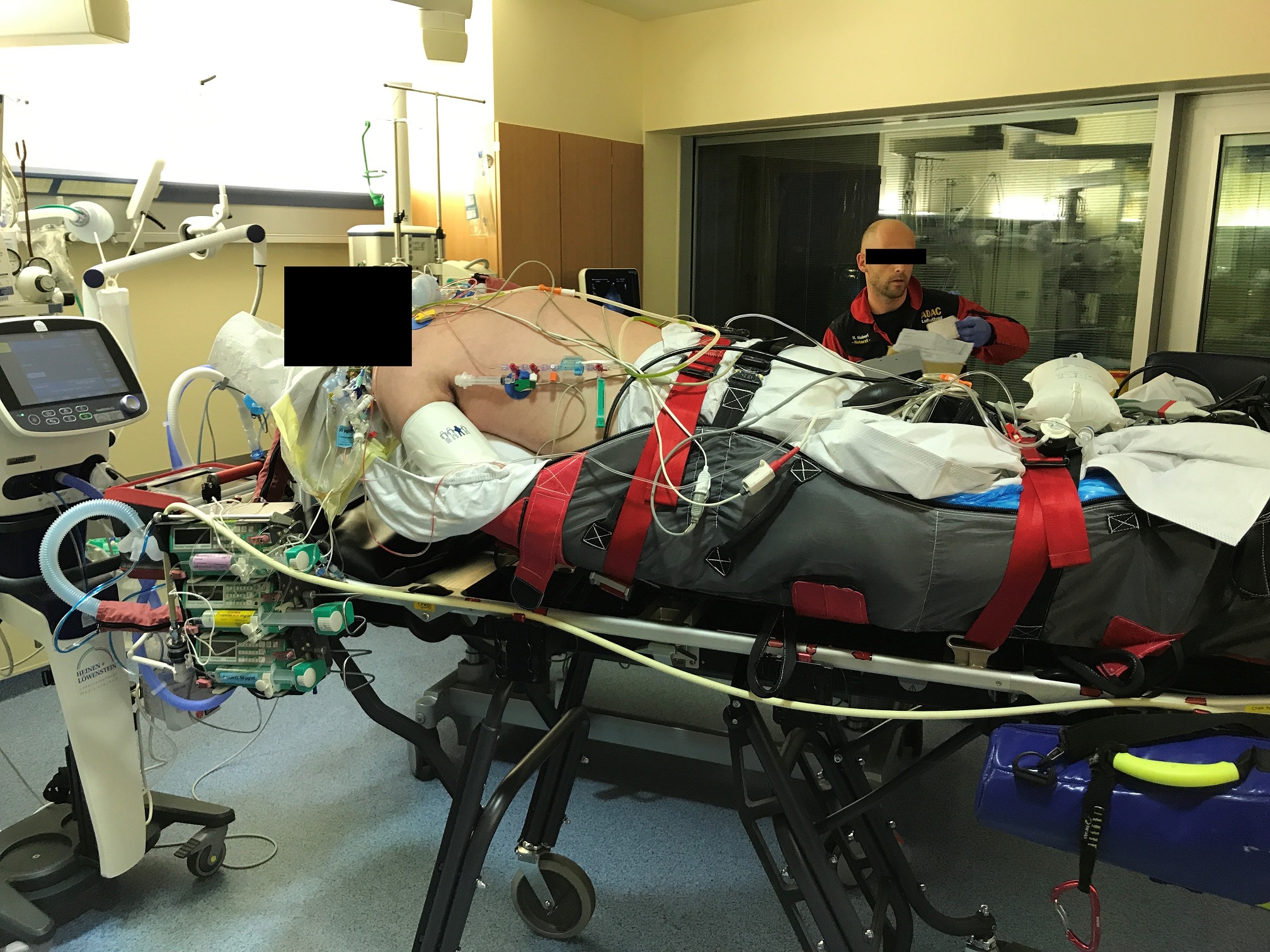
Simon Ilger and Helge Haberl of ADAC Air Rescue describe their experience of transporting a severely ill patient in need of ECMO to an ARDS center
Refractory hypoxemia continues to be a condition with a high mortality rate, with only a few therapeutic options being evidence-based. In the case of hypoxic respiratory failure with an expected mortality of more than 50 per cent, extracorporeal membrane oxygenation (ECMO) should be considered, according to the current criteria of the Extracorporeal Life Support Organization (ELSO), and with an expected mortality of 80 per cent or above, ECMO is indicated. The PaO2/FiO2 quotient and the Murray-Score are used for quantification. (1)
In most cases, ECMO is initiated by a team from an Acute Respiratory Distress Syndrome (ARDS) center in the peripheral hospital. This represents the best option in terms of patient safety, because on extracorporeal circulation, the respiratory situation stabilizes and the often-compromised hemodynamic situation also improves. The subsequent transport to the ARDS center for further treatment can then be carried out more safely.
History, clinical findings and therapy
A 47-year-old, 185-cm tall and 100-kg patient was initially admitted to a local hospital with a productive cough and fever. Apart from arterial hypertension, there were no comorbidities. The medical history was also unremarkable. Because of a suspected community-acquired pneumonia and corresponding infection markers (CRP 337 mg/l, leukocytes 3.1/pl, PCT 43.88 µg/l) a calculated antibiotic therapy with Ampicillin/Sulbactam and Clarithromycin had been started.
The patient’s respiratory situation deteriorated rapidly, so non-invasive ventilation became necessary. Hypoxic respiratory failure, refractory respiratory acidosis and increasing hemodynamic instability led to the patient being transferred to a nearby higher level of care hospital for further diagnosis and therapy. Endotracheal intubation was carried out as part of the transport preparations, which caused aspiration. Immediately after the transfer to the intensive care unit of the high care hospital, a bronchoscopy was performed that confirmed the aspiration. A bronchoalveolar lavage followed. Additionally, a chest X-ray showed diffuse blotchy shading in both lungs.
With a correspondingly reduced PaO2/FiO2 ratio (70.6) and after the exclusion of a relevant cardiac dysfunction, the diagnosis of ‘severe ARDS’ was made, according to the Berlin definition. The patient also suffered from septic shock, with an increasing need for vasopressors.
The initially calculated antibiotic therapy was continued. Sedation was difficult and initially carried out by continuous administration of propofol, fentanyl and S-ketamine. This regime was expanded to volatile sedation with isoflurane. Muscle relaxants were not administered. The patient was ventilated in a Biphasic Positive Airway Pressure Mode (BIPAP).
Following further deterioration of the respiratory situation, an escalation of the positive end-expiratory pressure (PEEP) and the peak pressure (Pinsp) was necessary. Finally, a PEEP of 14 mbar and a Pinsp of 30 mbar were administered. The ventilation rate was 18/min, with changing breathing time ratio (I:E) between 1:1.8 and 3:1. This allowed tidal volumes of 500ml and a minute volume between nine and 11l. An inspiratory oxygen fraction (FiO2) of 1.0 was also required.
With progressive hypoxia, hypercapnia and acidosis (07:21 hrs: pH 7.184, pCO2 76.1mmHG, pO2 70.6mmHG, FiO2 1.0), decision for positioning therapy in the form of a 135° position was made at noon (11:49 hrs: pH: 7.176, pCO2 78.4mmHG, pO2 62.7mmHG, FiO2 1.0). Because there was no improvement in gas exchange (17:00 hrs.: pH 7.158, pCO2 82.9mmHG, pO2 66.0mmHG, FiO2 1.0), by a Murray Score of 3.5, the decision to transfer the patient to an ARDS center was made. A university clinic about 100 km away confirmed they would accept the patient, but due to a lack of human resources, it was unable to provide a team for initiating ECMO therapy on site, so transfer by helicopter was organized.
Practicality and reality
However, initiation of ECMO therapy at the peripheral hospital is not always possible for logistical reasons. In these cases, ECMO therapy can only begin in the ARDS center, and it is necessary to transport these respiratory and hemodynamically unstable patients in conventional conditions.
According to current guidelines, prone positioning is typical for these patients and they are often prone at the time of the transfer request. Traditionally, the patient is transferred to a supine position before the start of transport, which often comes along with a subsequent deterioration of the gas exchange that had been compensated for in the prone position. The paradigm was that a critical patient could not be transported prone, because the supine position was considered necessary during the transport in order to be able to carry out further measures such as resuscitation.
Below, we report on the case of a 47-year-old patient with severe primary ARDS in the context of pneumococcal pneumonia who was referred to an ARDS center for ECMO therapy. Initiation of ECMO therapy on site could not be carried out, so conventional intensive care transport to the corresponding center was necessary. At maximal limited gas exchange, the team decided to perform the transport in prone position. The complication-free execution of the transport shows that proning patients can also be maintained in transportation if initiation of ECMO on site is not available.
Progress
A critical care helicopter (Airbus H145) with a crew consisting of pilot, co-pilot, emergency/critical care physician and critical care nurse/paramedic received the call at 19:25 hrs. The hospital was asked to supine the patient. In the pre-flight briefing, the medical crew expressed concerns about the safe feasibility of the transport due to the patient’s gas exchange being restricted to the maximum.
The flight departed at 19:44 hrs and arrived at the hospital at 20:10 hrs. Due to the instability of the patient, he had not been put in the supine position when the crew of the intensive care helicopter arrived in the intensive care unit; the patient was still in a 135° position. There was a sinus tachycardia at 144 bpm. A continuous intravenous catecholamine intake of 3.4 mg/h noradrenaline was required for hemodynamic stabilization. The peripheral oxygen saturation was only 65 per cent.
Despite the administration of muscle relaxants and a further escalation of ventilation invasiveness (Pinsp 32mbar, PEEP 15mbar, frequency 28/min., I: E 1: 1), no improvement in the respiratory situation could be achieved. The situation was rated as critical. First, the helicopter emergency physician reported back to the receiving university clinic and described the patient’s maximally critical situation. The indication for on-site initiation of ECMO therapy was considered as given by the receiving center, but this was still not possible. The entire crew then had a discussion about the feasibility of the transport under such conditions.
As part of the so-called FOR-DEC method, which is common in aviation, the decision was made to carry out the prone transport as follows:
Facts
Young patient with low comorbidities, currently with severe ARDS and urgent need for ECMO therapy. Initiation of ECMO therapy not possible on site. Remain on site with a presumably poor prognosis. Transport at the highest risk.
Options
- The patient remains on site.
- Helicopter transport in supine position.
- Helicopter transport in prone position.
- Transport by ground in a critical care ambulance.
Risks / benefits
- Not an option because the patient is likely to die.
- Risk of further deterioration after supining the patient with a high risk of further increase in hypoxia and possibly cardiac arrest in flight.
- Maintaining the prone position and maintaining the shortly compensated respiratory situation with the risk of not being able to react adequately to any further and possibly fatal deterioration (e.g. resuscitation).
- Significant time delay.

Decision
The only sensible option was option three. Without ECMO connection in a short time, the prognosis appeared poor. Immediate transport was required. The risk of complications in flight was estimated to be high; however, the gas exchange would be briefly compensated for with the patient in the prone position, so there was at least one option to reach the ECMO facility. Any further deterioration would lead to decompensation. In this context, rearrangement to the patient’s back did not seem feasible. The entire team clearly communicated that a sufficient resuscitation in the prone position was not feasible. Supining the patient in flight is also nearly impossible. The decision to carry out the transport under these conditions was made by consensus of the entire crew.
Execution
The colleagues of the delivering hospital and the receiving hospital were informed about the decision to carry out the transport in prone position and the transport preparation was started. First, the patient was prepared for the transfer to the stretcher. The sedation was deepened, the patient was moved from 135° to full prone position (180°) and the transfer was carried out gently on the stretcher using a vertical pull, with attention on avoiding critical pressure points and protecting the face. A ring to relieve the pressure on the face was made using cotton wool, and as far as possible, the upper extremities were placed next to the body, although the stretcher did not allow for the patient’s arms to stay by their head.
During the rearrangement, the patient mobilized large amounts of a thin, brownish pulmonary secretion. Endotracheal suction and the change of ventilation filter had to be performed. After this, the gas exchange deteriorated further (20.43 hrs: pH 7.144, pCO2 84.8mmHg, pO2 31.7mmHg, FiO2 1.0, SpO2 35 per cent). In order to rule out treatable causes, an orientating sonography of the pleura in the prone position was performed. Under difficult conditions, no new intrathoracic pathologies like pleural effusions or pneumothorax could be found. As ultima ratio, a Lachmann (recruitment) maneuver was carried out, which resulted in stabilization of oxygenation. A further escalation of ventilation (Pinsp 36mbar, PEEP 15mbar, frequency 28/min., I: E 1: 1) took place, as a result of which the peripheral oxygen saturation rose to 60 per cent. The helicopter monitoring equipment was then attached, and the syringe pumps with noradrenaline (3.4 mg/h), fentanyl (0.25 mg/h) and propofol (200 mg/h) were changed to those of the helicopter. The acidosis was treated by a weight-adjusted dosing of trishydroxymethylaminomethane (tromethamine). The subsequent change to the helicopter ventilator (Hamilton T1) was carried out without further complications, as was the transport to, and into, the helicopter. The helicopter departed shortly before 21:00 hrs, and the indicated arrival time at the university clinic was around 21:25 hrs. During the flight, the patient initially remained stable at a low level.
Monitoring showed:
- Sinus tachycardia at 150/min.
- Arterial blood pressure of 135/65 mmHg under noradrenaline 3.4 mg/h.
- End-tidal CO2 of 65mmHG.
- Peripheral saturation of around 65 per cent measured on the earlobe.
Ventilation was carried out in BIPAP mode with the parameters mentioned, which resulted in a tidal volume of around 400 ml and a minute ventilation of around 10 liters. During the flight, the SpO2 rose up to 80 per cent and the etCO2 decreased to 60 mmHg. An arterial blood gas analysis during the flight at 21:18 hrs showed the following: pH 7.225, pCO2 69.9mmHG, pO2 91.7mmHg, SO2 94.8 per cent. After an uncomplicated landing and ambulance transfer to the intensive care unit at the university clinic, the handover took place and the patient was transferred to the clinic, during which the patient was supined. After this maneuver, the gas exchange was again compromised in the form of a deterioration in peripheral saturation and a decrease in tidal volume. The reason for this is presumably supining the patient and the change of the respirator, which took place without PEEP loss due to clamping the tube. The veno-venous ECMO implantation immediately afterwards went smoothly. The patient’s condition improved promptly under the established measures for ARDS therapy.
Check
In hindsight, there were no major complications and the patient could be handed over to the target clinic in an improved condition.
Discussion
ECMO therapy is an integral part of rescue therapy for refractory hypoxemia in the context of severe ARDS. The prone position is one of the few established conservative measures for ARDS therapy with evidence. It is a grade A recommendation in patients with moderate to severe oxygenation disorder (Horowitz index <150) according to the German guideline: Lagerungstherapie und Frühmobilisation zur Prophylaxe oder Therapie von pulmonalen Funktionsstörungen (Positioning therapy and early mobilization for the prophylaxis or therapy of pulmonary dysfunction) of the Deutsche Gesellschaft für Anästhesie und Intensivmedizin (German society for Anesthesiology and Critical Care).(2)
The prone position in the sense of a 180° position – or the so-called incomplete prone position (135°-180°) – serves primarily to improve gas exchange and to mobilize secretions. The German guideline Invasive Beatmung und Einsatz extrakorporaler Verfahren bei akuter respiratorischer Insuffi zienz (Invasive ventilation and use of extracorporeal procedures for acute respiratory insufficiency) from 2017 describes the following effects of the prone position:
The change in respiratory mechanics.
The reduction in the pleural pressure gradient.
The reduction in tidal hyperinflation.
The reduction of ventilation-related damage to the lungs.(3-8)
This leads to a more homogeneous distribution of the breathing gases, a reduction in the ventilation-perfusion mismatch, and to the recruitment of non-ventilated lung areas.(9-12) The authors of the guideline do not describe absolute contraindications with appropriate risk / benefit assessment.(13)
Meta-analysis of randomized studies carried out showed a possible survival advantage due to the prone position in the patients who were proned early (<48 hours) after the onset of ARDS and/or in the long term (16 hours a day over seven days).(14,15)
Case reports and smaller cohort studies on transport under ECMO can be found in the literature, but data on transport in the prone position is very poor. There are only reports of two transports from Canada. In those reports. the authors implemented a special protocol in this case.(16) Our case shows that transportation of a patient in prone position with severe ARDS is an option if ECMO therapy is not available on site. It is not necessary to place the patient supine, and the associated risks and frequent deterioration of the condition can be avoided.
However, it also shows that this type of transport can only represent an ultima ratio, since the possibilities of intervention are very limited and, for example, moving the patient onto their back in the helicopter cabin seems almost impossible. Furthermore, a high level of expertise in the care of ARDS patients in the prone position is necessary, which in this case existed on the part of both the accompanying critical care physician and the critical care nurse/paramedic. In any case, an individual risk / benefit assessment must be carried out; ideally, the patient’s relatives should be fully informed. The improvement in gas exchange in the case described is an interesting observation. Presumably it has multifactorial causes: on the one hand the further escalation of ventilation, and on the other hand the mobilization, as well as the vibrations associated with the helicopter flight, could have led to secretolysis and recruitment of less ventilated lung areas.
Conclusion
Transport of a critically ill patient in a prone position is possible, but it is a great challenge for the team and requires an intensive risk / benefit assessment, as well as appropriate expertise, and should only be considered if safer alternatives such as the initiation of ECMO therapy cannot be implemented on site.